Kyoto, Japan -- Toward the right side of the periodic table below oxygen you'll find the chalcogens, or "ore-forming" elements. The chalcogens that occur naturally, including sulfur, selenium, and tellurium, are all somehow involved in biological processes. Molecules containing sulfur, like the antioxidant glutathione, play a central role in redox regulation, the balance between oxidation and reduction that is essential for maintaining cellular health.
Recent studies have suggested that the heavier selenium and tellurium are active in biological redox systems as well, but the instability of molecules containing chains of different chalcogen atoms has made structural analysis difficult. Traditional methods have largely relied on mass spectrometry, which cannot be used to directly observe molecular bonds. This limitation motivated a team of researchers at Kyoto University to develop a method that would allow them to more clearly observe chains of chalcogens.
"We have long been interested in understanding how subtle atomic substitutions can alter biological function," says corresponding author Kazuma Murakami. "Chalcogen chemistry offers a unique window into redox biology that remains largely unexplored."
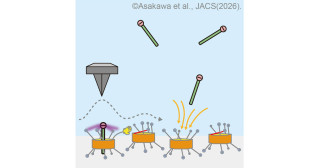
The team's newly developed method involves an in situ reaction performed in an aqueous solution of oxidized glutathione-cystine molecules, into which atoms of either selenium or tellurium are inserted. They then use 1H-detected 77Se/125Te nuclear magnetic resonance spectroscopy, NMR, to analyze the molecule structure. Finally, they assess the redox activity with radical scavengers, substances that protect cells from damage caused by unstable molecules while also preventing ferroptosis, a type of controlled cell death.
This innovative approach enabled the researchers to generate and characterize heterologous trichalcogenide molecules containing sulfur, selenium, or tellurium, allowing them to directly observe the unstable bonds between different chalcogen atoms. Their observations revealed that these compounds exhibit strong redox activity.
"This is the first direct spectroscopic view of heterochalcogen bonds in redox systems," says Murakami. "By combining multinuclear NMR with superchalcogenide chemistry, we have opened a new avenue for studying redox-active biomolecules."
The new method used in this study has the potential to enable rational design of novel redox-active molecules and support the development of functional biomolecules and peptides. It may also contribute to research involving oxidative stress and diseases resulting from ferroptosis.
Next, the team plans to expand their new method to more complex biomolecules and to continue to explore the biological roles of chalcogen-modified glutathione derivatives. They also hope to design novel redox-active compounds that have potential use in medical therapies.

【DOI】
https://doi.org/10.1021/acsmeasuresciau.5c00193
【KURENAI ACCESS URL】
http://hdl.handle.net/2433/300317
Kazuma Murakami, Keisuke Tao-Kakuyama, Thi Hong Van Nguyen, Katsutoshi Nishino, Takaaki Akaike (2026). Exploring Chalcogen Connection in Glutathione Trichalcogenides via ¹H-Detected ⁷⁷Se/¹²⁵Te NMR. ACS Measurement Science Au, 6, 2, 466-475.