Analyzing catalytic activity with operando X‑ray Absorption Spectroscopy
In a collaborative research initiative lead by Professor Tsunehiro Tanaka and Dr. Hiroyuki Asakura of ESICB Kyoto University, researchers in Japan have gained further insight into the dynamic behavior of the precious metals used in devices such as catalytic converters, opening the door for manufactures to build more durable and efficient converters. The findings were published in the Journal of the American Chemical Society .
Essential components of these converters -- known as three-way catalysts, or TWCs -- are the Platinum group metals, or PGMs. These elements include Rhodium (Rh), Palladium (Pd), and Platinum (Pt) and are vital for the purification of harmful gases in automobile exhaust such as nitrogen oxides (NOx), carbon monoxide (CO), and hydrocarbons (HCs).
However, since these devices are subjected to intense heat over a long period of time, the metal compounds eventually fuse, reducing surface area and lowering efficiency. Coupled with rising global demand for automobiles and the economic constraints of PGMs, an extensive number of studies have been performed on the mechanism of catalysis, degradation, and regeneration of TWCs.
To better understand the molecular machinery in TWCs, the team studied the behavior of a species of catalysts that use Rhodium (Rh) as its active metal. The researchers constructed an operando X-ray absorption spectroscopy (XAS) system equipped with a micro gas chromatograph, a NOx meter, and a quadrupole mass spectrometer.
O perando is a generic term describing the application of spectroscopic techniques to materials to elucidate their function, structure, and electronic states. These combined techniques have generated unprecedented and real-time insights into the molecular behavior of Rh species catalysts.
The team found that Rh reacts differently when under oxidative or reductive conditions. In the oxidation process it was observed that the oxidized points gradually grew and expanded on the surface. In contrast, during the reduction process, the metallic state was reduced swiftly and randomly.
The combined data present new findings of altering surface rearrangement behavior for Rh catalysts.
The next step in the team's research will be to more closely reproduce the high temperature conditions that TWCs normally endure. Overall, these findings have validated the benefits of operando XAS to study the properties of catalytic metals, and the team hopes that their tools and methods will be employed in the study of other materials in the future.
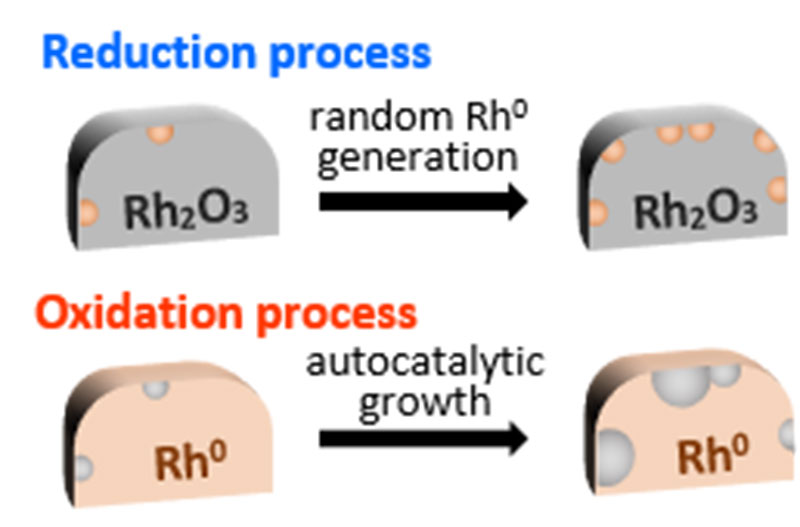
Redox behavior of Rh species when the gas composition is switched (Kyoto University/Tanaka Lab)
Paper information
【DOI】 https://doi.org/10.1021/jacs.7b07114
【KURENA ACCESS URL】 http://hdl.handle.net/2433/229120
Hiroyuki Asakura, Saburo Hosokawa, Toshiaki Ina, Kazuo Kato, Kiyofumi Nitta, Kei Uera, Tomoya Uruga, Hiroki Miura, Tetsuya Shishido, Junya Ohyama, Atsushi Satsuma, Katsutoshi Sato, Akira Yamamoto, Satoshi Hinokuma, Hiroshi Yoshida, Masato Machida, Seiji Yamazoe, Tatsuya Tsukuda, Kentaro Teramura, and Tsunehiro Tanaka (2018). Dynamic Behavior of Rh Species in Rh/Al2O3 Model Catalyst during Three-Way Catalytic Reaction: An Operando X-ray Absorption Spectroscopy Study. Journal of the American Chemical Society, 140(1), 176-184.