December 25, 2013
A Kyoto University research team has used iPS cells generated from the cells of patients with FOP (fibrodysplasia ossificans progressiva) to successfully replicate the pathological conditions of the disease. The team members included Associate Professor Makoto Ikeya (Kyoto University, CiRA), Professor Junya Toguchida (Kyoto University, CiRA/Institute of Frontier Medical Sciences/Graduate School of Medicine), postgraduate student Yoshihisa Matsumoto (Kyoto University, CiRA/Institute of Frontier Medical Sciences; Nagoya City University Graduate School), and Dr. Edward C. Hsiao (Gladstone Institutes/UCSF).
The research findings were published on December 9, 2013, in the Orphanet Journal of Rare Diseases.
Background
FOP is a disease in which bone gradually forms within muscle, tendon, ligament and other soft tissues. Affecting around one in two million people, it is one of a group of rare and intractable illnesses sometimes known as orphan diseases . Research so far has established that FOP is caused by a mutation in a gene known as ACVR1 which causes it to become overactive. However, because taking tissue samples from FOP patients promotes the ossification process and because of the limitations of mouse models of the disease, the detailed mechanism of FOP onset has remained unclear.
Although scientists had succeeded in generating iPS cells using cells taken from FOP patients, no assessment had previously been made of the differentiation potential of these iPS cells. Having established a method of inducing iPS cells to differentiate into bone and cartilage, Professor Toguchida and his team investigated the ability of these FOP patient-derived iPS cells to similarly differentiate into bone and cartilage. The cells used in their research to generate iPS cells were from a bank of cells established using dermal fibroblasts from five FOP patients.
Research Result
FOP patient cells and a set of control cells were sourced from a dermal fibroblast bank and used to prepare four lines of FOP patient-derived iPS cells and two lines of control iPS cells. All of these cell lines were pluripotent cells with the ability to differentiate into cells of all three germ layers cells . When these cells were cultured for 15 days in conditions promoting bone formation, ossification was more advanced in the FOP patient-derived cells than in the control cells (Fig. 1).
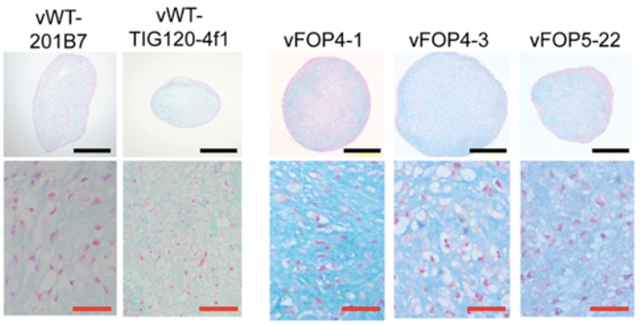
Fig. 1: When iPS cells were cultured in an environment promoting bone formation, the ossification process was enhanced in FOP patient-derived cells.
iPS cells were cultured in an environment promoting differentiation into bone and the extent of ossification was measured on days 1, 6, and 15 by von Kossa staining. On day 15, black staining was evident which was more intense in the FOP patient-derived iPS cells, indicating more advanced ossification.
The researchers succeeded in partially replicating the pathology of FOP outside the body, thus establishing a model with which to evaluate candidate pharmaceutical substances for their ability to inhibit the process of conversion to bone (ossification) or cartilage (chondrification). This achievement is expected to accelerate research into drugs for FOP. The research also indicates the possibility of using iPS cell technology to replicate the pathology not only of FOP, but also of other rare diseases in which it is difficult to obtain research samples from patients. It also demonstrates that iPS cell technology has a role to play in drug discovery research and in the elucidation of disease mechanisms.
Paper Information
Paper Title
"Induced pluripotent stem cells from patients with human fibrodysplasia ossificans progressiva show increased mineralization and cartilage formation"
[DOI] http://dx.doi.org/10.1186/1750-1172-8-190
Orphanet Journal of Rare Diseases
Published: 9 December 2013, 8:190
Authors
Yoshihisa Matsumoto, Yohei Hayashi, Christopher R Schlieve, Makoto Ikeya, Hannah Kim, Trieu D Nguyen, Salma Sami, Shiro Baba, Emilie Barruet, Akira Nasu, Isao Asaka, Takanobu Otsuka, Shinya Yamanaka, Bruce R Conklin, Junya Toguchida, and Edward C Hsiao

